Megan B.
Great experience from start to finish everyone was so nice and made me feel comfortable. SEE MORE
I have been doing breast augmentation in private practice since 1996. During that time, I have developed a list of important issues that patients need to know about breast augmentation and implant choices in Salinas, CA.
Breast augmentation, also known as augmentation mammoplasty, is a surgical procedure used to increase the size and projection of the breasts. This breast procedure involves the creation of incisions and the placement of breast implants beneath the tissues.
The best breast augmentation candidates are in good overall health, as this reduces the likelihood of complications. Good candidates are non-smokers, since smoking negatively affects the health of the skin and can result in a prolonged healing process.
Additionally, ideal breast enlargement candidates have realistic expectations for their results. The best way to have clear and realistic expectations is by looking over breast augmentation before and after photos.
The first decision that most women who are considering breast augmentation need to make is what type of implant to use. Some prefer large breast implants, whereas others are more suited for small breast implants. Deciding on the best breast implants for you requires learning more about your implant options.
I tell each of my potential breast augmentation patients the following:
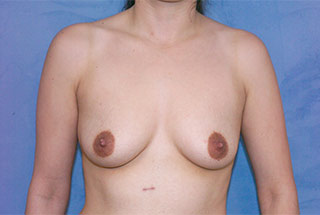
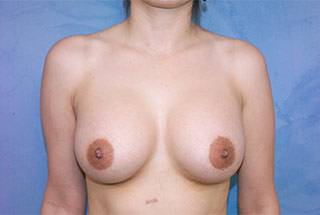
* Individual results may vary.
View before-and-after pictures of real patients of Dr. Matthew Romans
Saline
Silicone
There are now silicone gel implants available that have a very viscous gel material in the center (the “gummy bear” implant). They originally came only as shaped and textured implants but Allergan introduced round smooth cohesive gel implants in 2017. The gel material is very cohesive so the center is “less liquid”.
The cohesive gel round implants hold their shape better so there is some consideration that they may have less rippling. When gummy bear implants experience a rupture, they do not leak; instead, their material stays safely inside the implant. They are more expensive than traditional gel implants and are not for everyone.
There are three types of Allergan gel smooth implants; the “traditional gel”, the “soft touch”, and the full “cohesive gel”.
Type 1. Natrelle INSPIRA® Responsive. Least amount of cross-linking. Has softer, more-natural appearing upper pole result. This is the least expensive of the Natrelle INSPIRA® product line at about $800 each.

Type 2. Natrelle INSPIRA® SoftTouch. Has more cross-linking between molecules. We feel this offers the best combination of upper-pole fullness, price and gel stability at about $875 each. This is the most common implant we use at Salinas Valley Plastic Surgery. We have it in stock at the Surgery Center on the shelf for most sizes we use so the implants are not special order for your surgery.

Type 3. Natrelle INSPIRA® Cohesive. This is the full “gummy bear” smooth gel implant. It gives the most upper pole fullness and is slightly firmer than other silicone implants. This implant is the most expensive of the Natrelle INSPIRA® implant line at about $1175 each.

Type 4. INSPIRA®Textured Shaped cohesive gel. This is the original gummy bear implant approved in the United States. At Salinas Valley Plastic Surgery we have used this implant commonly for breast reconstruction, but only occasionally for cosmetic augmentation. As of mid-2018, we have stopped using these because of concerns outlined below about BIA-ALCL.

Information from the website of the FDA.
Information from the ASPS (American Society of Plastic Surgeons).
It is possible to combine breast enlargement with a breast lift in order to create more comprehensive enhancement and rejuvenation. This combination breast procedure is known as a breast augmentation with lift, or augmentation mastopexy.
Pairing a breast lift and implants reduces sagging while increasing volume and projection. A breast lift without implants is also available if you are purely interested in resolving sagging.
Delivery of anesthesia is the first step of any breast surgery. Usually, general anesthesia is chosen, but in some cases, local anesthesia with sedation may be used. This choice will be made during the initial consultation. Anesthesia keeps the patient comfortable and free of any pain during the surgery.
Following this step, incisions are made. One of the following incision options may be used:
Pockets are then made in the breast tissues through the incisions. The breast implants are placed into the pockets snugly to prevent implant migration. After the implants are positioned correctly, the incisions are closed with medical sutures and then dressed appropriately.
It is important to avoid rushing the breast augmentation healing process; the body has gone through a change and needs time to recover properly. During breast augmentation recovery, you will need to follow your surgeon’s post-surgical care instructions very closely.
You will need to address bruising and swelling in the surgical area using a cold compress. Wearing a post-surgical bra without underwire will help to support the tissues as they heal.
Taking a week off from work will be necessary, as will avoiding any sort of strenuous activity for several weeks. This includes sports and physically taxing exercise regimens.
When considering breast enlargement, patients typically put price high up on their list of potential concerns. Breast augmentation cost is determined by a multitude of factors, include the length of time required to complete the surgery, the breast implants chosen, and the specific techniques used.
Patients who are interested in breast augmentation in Salinas, CA, should contact Salinas Valley Plastic Surgery for a consultation. During a consultation, patients can view breast augmentation before and after photos, ask questions, and more. Take the first step towards achieving your aesthetic goals – make your appointment today.

Get the care you need
For over 30 years, CareCredit has been providing a valuable financing option for treatments and procedures that typically are not covered by insurance, or for times when insurance doesn't cover the full amount. CareCredit is also used by cardholders to pay for deductibles and co-payments.

5.0 rating out of 148 reviews
For informational purposes only, a link to the federal Centers for Medicare and Medicaid Services (CMS) Open Payments web page is provided here. The federal Physician Payments Sunshine Act requires that detailed information about payment and other payments of value worth over ten dollars ($10) from manufacturers of drugs, medical devices, and biologics to physicians and teaching hospitals be made available to the public.
The Open Payments database is a federal tool used to search payments made by drug and device companies to physicians and teaching hospitals. It can be found at https://openpaymentsdata.cms.gov.